Previously I wrote up why the 9V battery sucks. As I thought more about that post, I realized, I never explained how much energy is in a 9 V battery versus say a couple of AA batteries.
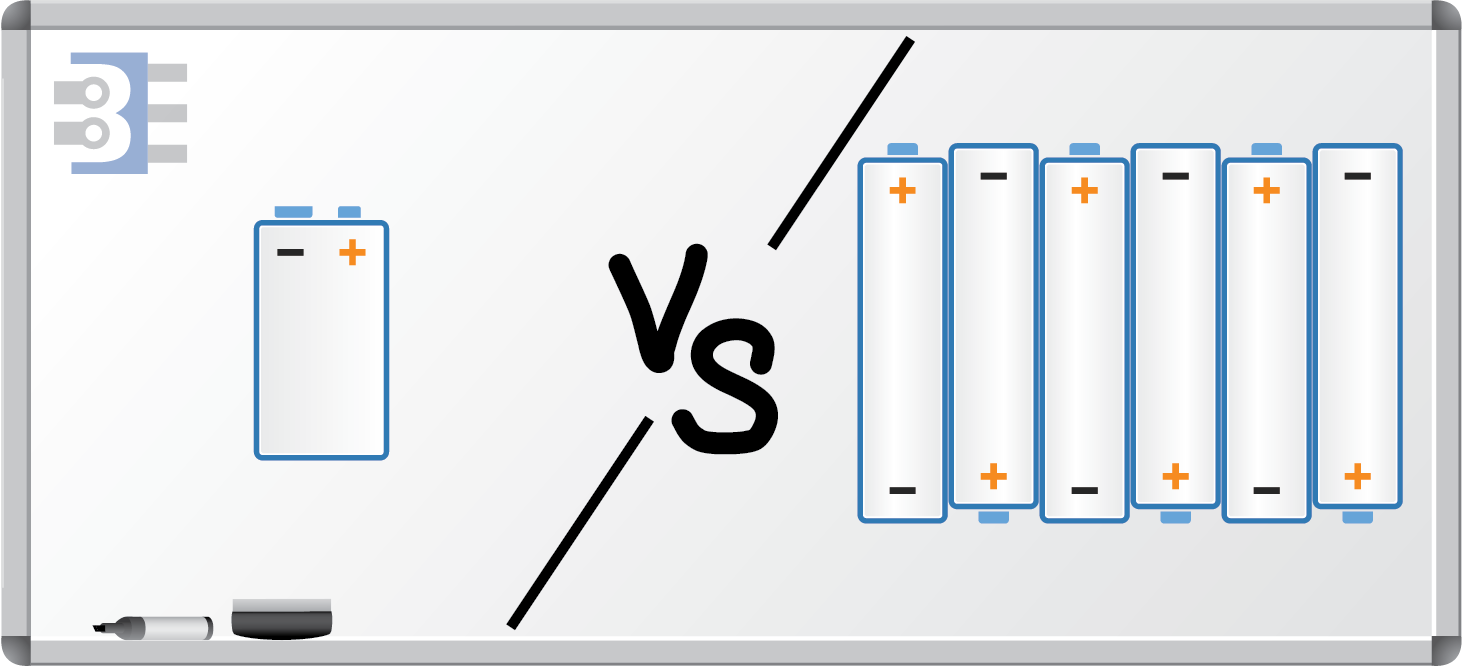
For this post, I am going to break down the energy stored in a 9 V battery, the small rectangular kind and compare it to what you get with 6 AA batteries. Yes, it takes up a little more space, but you might be surprised by the difference.
Power vs. Energy
While 9 V sounds impressive, voltage is not the only consideration when it comes to the “power.” Or, more correctly, the energy in a battery. To start, let’s make sure we understand the difference between energy and power.
Power
While the term “power” does tend to get misused, it has a very scientific definition. Power is “the rate at which energy is delivered to a circuit.” Power’s SI unit is “Watt.” A watt is defined as one joule per second.
Properties of a battery, like its internal resistance and how that resistance changes with temperature, determines the battery’s maximum deliverable power. Now, let’s compare that to the term Energy.
Energy
The lesson I remember from high school physics is that energy can not be created or destroyed. You can, however, transfer or transform it. In the case of a battery’s cells, energy is stored chemically and delivered electrically. This distinction is important: cells convert energy between chemical and electrical forms.
You can waste, consume, or store energy. But you cannot create or destroy it. (Apparently, you can also buy small cans of it marked as Red Bull.)
The SI unit joule (J) expresses the unit used to measure energy.
Energy Stored
The energy capacity of a battery is how much energy it can store in its chemical structure. We can calculate a battery’s stored energy with a simple formula.
Calculating Energy in a 9V Battery
The formula calculates the energy stored in a battery:
\(E = V \times I \times t\)In this formula “V” is the (nominal) voltage of the battery, “I” is the current capability in amps, and “t” is the time in seconds.
From my previous 9V battery post, we know that a typical alkaline battery stores 600mAh, at best. That is part of our current capability. However, that rating is in milliamp-Hours, which is a funny unit of measure. To convert to amps per second, just multiply the number by 3600.
With these three pieces of data, our equation becomes:
\(E=9V\times600mA\times3600s=19440J\)19,400 joules sounds like a bunch, right? Next, let’s compare to the amount of energy in a single AA battery.
Calculating Energy in an AA Battery
A single Energizer AA, or E91, is rated for just under 3000 mAh at 25 mA drain. For this calculation, I picked 25mA because that is the nominal current draw for a 9 V. As a conservative value, let’s use 2750 mAh.
Using the same energy formula before, we can calculate the amount of energy in a single 1.5 V AA battery.
\(E=1.5V\times2750mA\times3600s=14850J\)Wow, that is almost the same number, yet, the battery’s rating is only 1.5 volts! That is pretty amazing when you think about it! Of course, We need six more cells to get to 9 V. Which gives the total energy of:
\(E_{6}=14850J\times6Cells=89100J\)Obviously, that calculates as 6X a single AA and just slightly more than 4X a 9V battery. Next, let’s take volume into account.
Energy Density
Using the volume and the weight of the battery, we can calculate the energy density of both the 9V and AA.
We can get those values from Energizer’s data sheets for Zinc / Manganese-Dioxide (MnO2), aka Alkaline, batteries. Combine that and we can get the energy densities.
[table] Battery,Energy (J),Weight (g),Energy / Gram,Volume (cm3),Energy / cm39V,”19,440″,45.6,426.3,21.1,921.3
Single AA,”14,850″,23.0,645.7,8.1,1833.3
Qty 6 AA,”89,100″,138.0,Same,48.6,Same
[/table]
So for three times the weight, you get 4.6 times the available energy, while only taking up about 2x the space required.
Conclusion
As I said in my previous 9 V battery post, don’t be lulled into the myth that “9Vs Have More Power!11!.” When you do the math, their energy density is pretty lousy compared to the little 1.5 V cell.
If you can afford the space, you are going to be much happier with the energy provided by six AA batteries, compared to a single 9V battery.
References
- Energizer AA (E91) Datasheet
- Energizer 9V (522) Datasheet
- Battery Basics from Gears Educational Systems
- Excellent “next level” information, exercises, and measurements




30 Comments
When you multiply Ampere-hours by 3600 seconds/hour, the units of the result is Ampere-seconds — not Amps/second.
I’ve been trying to improve my lawn for the last couple of years and bought a Scotts Wizz battery operated seed spreader (twice!) (https://shrtm.nu/0FMe). The first unit I got was just a dud. it would continue working even after I stopped pressing the trigger. I then got another one of the same which worked fine last summer but this year when I went to use it it seemed pretty anemic. I thought the batteries were just old but after replacing them with fresh ones (twice!) it would work perfectly for a say 5 minutes then slow down as if it was struggling. I have come to the conclusion that this spreader is just mediocre design. I have a feeling that when I hosed it down last summer some water/mud/debris got lodged somewhere in the insides.
So my question is: do you think the electric motor can tolerate 9 volts? I’m thinking I can but one of those six AA battery holders and sort of just tape it to the outside. If I get a couple of more years use out of this poorly designed spreading it might be worth it.
Thanks!
Thanks for this. I just bought a multimeter which uses 2AA batteries. I suspected it would have more energy than my previous one with a 9V battery, and you have proved it for me.
Been using both for more than 50 years. 9-volts can lie around for years and–no leaks.
Just a minor point. In your energy calculation, you are actually multiplying by 3.6 instead of 3600. This is to convert mili-amps into amps, as well as converting hours into seconds. Your results are correct, but I thought the current conversion deserved a mention. Thanks, great article.
I have an older guitar which includes a 6AA battery pack which is attached using a 9v connector. I realize that simply substituting a 9V battery will not work (for very long). But is there a way to replace the 6AAs with a Li-ion battery pack that would provide higher energy, longer life and the ability to recharge? And taking up the same space as the 6AA pack.
I thought because the AA batteries were in series in order to get 9v that your amp hr is equal to 1 cell,so technicaly it has less reserve than a 9 volt battery
In this post, I am talking about energy which is measured in Joules. The total amount of energy in a bank “adds up.” The 1.5volt cells used to make up a 9V PP3 battery are significantly smaller than a single AA cell. Which means the total energy in the package is going to be much smaller than 6 AA cells combined.
That said, you are correct. Milliamp-hours do not add up when batteries are placed in series. However, that is a measure of Power. Remember that power is energy expended over time, aka work. The power delivery capability of a PP3 9V battery at 25 mA is about 500 milliamp-hours. While a LR6/AA’s capacity if over 2500 milliamp-hours. Put 6 AAs in series and their power delivery is still 2500 milliamp-hours.
Hello. I found you when I googled re: “What is the output of a 9v battery in ma”. I wanted to ask a question, and them I read some of your posts. So, I’m in electronics pre-school an you’re Nicola Tesla. Anyway, I started this, so I will ask while I remain prostrated before you and avoid eye contact.
I play electric guitar with 9 volt effects pedals. They are very small “nano” pedals that draw between 10ma and 30ma each.
I daisy chain 6 of these pedals together on my pedal board.
I just bought five 9v battery boxes, online, with leads and on/off switches for about 60cents each.
Is there a way I can wire the boxes together to maintain 9V, and increase the life of my “power bank” by 5X?
Now, I will humbly retreat from your presence on hands and knees, ever facing your magnificence, with my head bowed in awe and respect.
Yours truly,
Don
Batteries in parallel will charge-discharge each other, especially when the load is light. You need balancing resistors to help prevent this charge/discharge cycle. If you’re going to “bank” 5 PP3 9Vs, you’re better off putting 6 AA cells in series. The power lost from the 9V batteries in parallel easily offset the slightly higher amount of energy they offer over 6 AAs in series.
Just a little FYI to the other comments you got here…. Most batteries stop working when their voltages drop below a certain percentage as they tend to do as they get used and a lot of energy gets wasted as a result. The electronics stop working as the voltage gets too low, and even though they contain 20% or so of their “full” energy, you end up having to replace them. There is a circuit nicknamed a “joule thief” (google it) that will keep the voltage at nominal and increase the life of your batteries. Sometimes this, or a similar circuit exists in the electronics of the device, and you can probably buy a chip or little board that will do this work for you. If you are using one battery box for multiple devices, like 6 AA batteries, it might be worth your while to add such a circuit to your battery box. This will keep the voltage constant at 9V as the batteries get used, and thus increase the life of your battery box. Just something to note if you are into building electronics or have a friend that does….:-)
Nice calc. You saved me some time. My son is making a hovercraft for science project. the kids were connecting 9Vs together to get enough amps to run the electric motors. I think 6 AA will work much better than 3-4 9V. the rules say they have to have 9V max alkaline. So the weight and power this is probably the best solution. Using larger C or D batteries will weigh it down. It only has to run for 15-20 sec at a time and carry up to 2kg–I don’t think that would be possible for kids unless I helped a lot!
No doubt AAs or Cs are the way to go. The “9V max” might be a red herring if the contest/project was set up correctly! Send some pictures (you can email them to james at my domain name.)
Thanks for your reply. It helps!
The robot I’m making now is kind of compact, and I’m trying to think how I can fit all the things together like motors, sensors, circuits, batteries, etc. into a robot. Because of that, I might want to use 6 AAA batteries to power up the NXT instead of using AAs because the batteries are small, but I don’t understand about batteries so much, so to make sure I’m not doing anything wrong, I have a question for that.
Is it OK to connect 6 AAA batteries to NXT? (What’s the differences between using 6 AAA batteries and 6 AA batteries to power the NXT?)
AAA’s have slightly higher ESR (means more power loss) and less capacity than an AA. They’re still a better option than a 9V battery.
Most 9V batteries are now just 6 AAAA alkaline cylinder batteries (actually they are slightly shorter than true AAAA) arranged in the familiar rectangular package. So it’s not surprising that 6 AAAA batteries have less energy than 6 AAA or 6 AA. Both Energizer and Duracell do this, at least in the batteries I just unpeeled.
The differences in energy/gm or /cm3 are mostly based on the amount metal used in the six tiny battery tubes, and the air-space around them as they are packed in the 9V box.
Just a dummy here. I made a portable phone charger using a 9volt battery and it is currently charging my phone. I hope. Not much progress, yet, but I’m patient. So if I made one using the same amount of space with two AA batteries, trying to understand energy vs power here, does this mean the 2 AAs would have more energy to quickly charge the phone even though they would only have a fraction of the power to do the work? Perhaps meaning they would have more energy to provide more power, thus, turning their lower power voltage into a higher power due to higher energy, thus, charge my phone faster? So.. energy = power amplification, turning low power into high power? This is assuming I understood correctly that power = voltage, energy = something. Not sure… watts? No, that’s voltage isn’t it? Not sure, not sure…not…. sure….
Energy is the ability to do work. Power is the actual work. The subtle difference between the two is time. If I tell you a battery has 14,850 J of energy, the missing component is how fast can you use that energy. That’s where “power” comes in. The faster you draw current out of a battery, the more power you waste on its internal resistance. So part of the energy that is stored gets wasted. Which is one reason why the mAh (milliamp-hour, notice the time factor) rating of a 9 V is so low compared to a AA.
I don’t like using the term “quickly” because it is vague.
If you want to quickly charge your phone, that means drawing higher current out of a battery. You’ll get more total energy from a AA, because the battery is wasting less of that energy on itself. You would also have to take into account the losses from a boost circuit that boosts up the voltage. Of course, then you also have to account for the much higher current draw. (Boost circuits draw more current than they put out, to achieve more voltage than they get in.)
I haven’t done the math, so I can’t tell for sure the best option. My guess is two AAs with a boost circuit is going to offer more total energy than a single 9V with a buck, or (gasp) a linear regulator.
One last question,
If I decide to use 4 x AA batteries for my Amplifier, does it matter whether I put them in series (6 volts @ 2K mAh) and buck the voltage down to 5 volts or I put them in parallel (1.5 volts @ 6K mAh) and Boost the voltage to 5 volts? or lastly put two in series and then the rest parallel giving me (3 Volts @ 2K mAh + 1.5 Volts @ 4K mAh) The efficiency of the converter is at 90%.
The outcome I would like to know is which setup, series, parallel, or combo will provide the most longevity if there are any with these setup?
Thank you!
I would just be walking through the same math. Look at current in vs current out when you boost the 1.5 to 5v. Then look at the same chart and see how the mAh rating would change.
Hi,
Lets say you have a buck/boost converter with a hypothetical 100% efficiency for simplicity. If you were to step down a 9V battery using the converter to 1.5 volts, then would the 9V have the capacity equivalent or a little greater than that of a single 1.5 AA battery? I’m using the typical capacity you’ve mentioned in your blog for each battery type.
The reason why I ask is I’m trying to decide if I should power a mini amplifier using 2 AA battery boosted to 5 volts or use a single 9V battery bucked to 5 volts. 90% conversion efficiency.
Some more info, I’m powering a PAM8403 amplifier that requires 5V. Measured current Draw was like 40mA but I was told it could exceed up to 100mA. Thank you.
Thank you.
[Combined your comments. -James]
You’re on to a good idea. Let’s look at numbers.
5V @ 100mA (100% eff) out, would be 55mA at 9V, in. Consider inefficiencies to make it closer to 60mA. Now look at the discharge chart from the Energizer datasheet (linked in the references above.)
When discharging at 25mA the battery’s capacity is rated at 600mAh. At 100mA it drops to about 500mAh. So your example’s current draw going from 100mA to 60mA isn’t going to see a significant rise in capacity. Now if the 100mA is a peak value, then you need to consider that as well. However, I would say from 40mA to 60mA draw, the battery life is going to depend more on temperature than load’s current.
Compared to 2 AAs, at 100mA draw they have approximately 2500mAh. If you boost your 100mA to 5V @ 90%, you’re at 167mA. Rounding up to 250mA, they provide about 1900mAh. So about 4 times the capacity of the 9V. For reference, the even smaller AAAs would be around 700-800mAh under these conditions. Still more than a 9V.
Thank you for your response!
If it is one of those cheap chinese converters then it’s probably never reaches that 90%, also, converters doesn’t have just one efficiency, depending of the quality and design converters are better at converting different ranges of voltage
Have you also done a price comparison between 1 9v and 6 AA’s of the same manufacturer?
Sure. A single 9V costs less than 6 AAs. But the cost per joule is different. The 9V runs about $0.16 per kJ. While the 6 AAs would cost $0.13 per kJ. Base price used a two-pack of 9V and a two-pack of AAs on Amazon.
Ulrich wrote me asking…
A conservative value means “cautious” or “low estimate.” Reading precise values on the AA data sheet is difficult. So to not over represent the data, I used a smaller number for the capacity. In engineering we often call this cautious estimation “being conservative.”
The answer depends on the intended application. When designing a circuit, you need to decide what is important. The reason for my comparison is to point out that just because the battery has more volts it actually has very little energy available.
The key factor here is “If you can afford the space” (and weight) of the AA batteries in your project. Also, did you include the total volume including the battery holder that 6 x AA batteries require?
If you dismantle a 9V battery inside it is just a collection of 6 tiny cylindrical cells so of course it is going to have less energy available. Moreover, as you reduce the size of a battery it’s energy efficiency will decrease (though I haven’t calculated that)
I think that you should repeat this thought experiment using 6 x AAA and 6 x AAAA batteries to give a more reasonable comparison of volume and weight
I have two questions.
1. I use NXT lego mindstorms to make robots. If anybody knows or have used NXT lego mindstorms before, would it be OK to use one 9v battery instead of 6 AAs? If I use one 9v battery for NXT, would it damage it?
2. How can I post a photo on this comment? Because I might want to ask questions by posting photos.
No, using a little 9V battery won’t “damage” the NXT. However, it won’t work very well. This post goes into more detail on why you shouldn’t use 9 volt batteries for anything with motors, LEDs, or microcontrollers: https://www.baldengineer.com/9v-batteries-suck.html. As for posting pictures, the best option is to post them on something like imgur and include the URL.